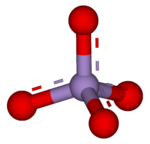
Active Mixed‐Valent MnOx Water Oxidation Catalysts through Partial Oxidation (Corrosion) of Nanostructured MnO Particles - Indra - 2013 - Angewandte Chemie International Edition - Wiley Online Library
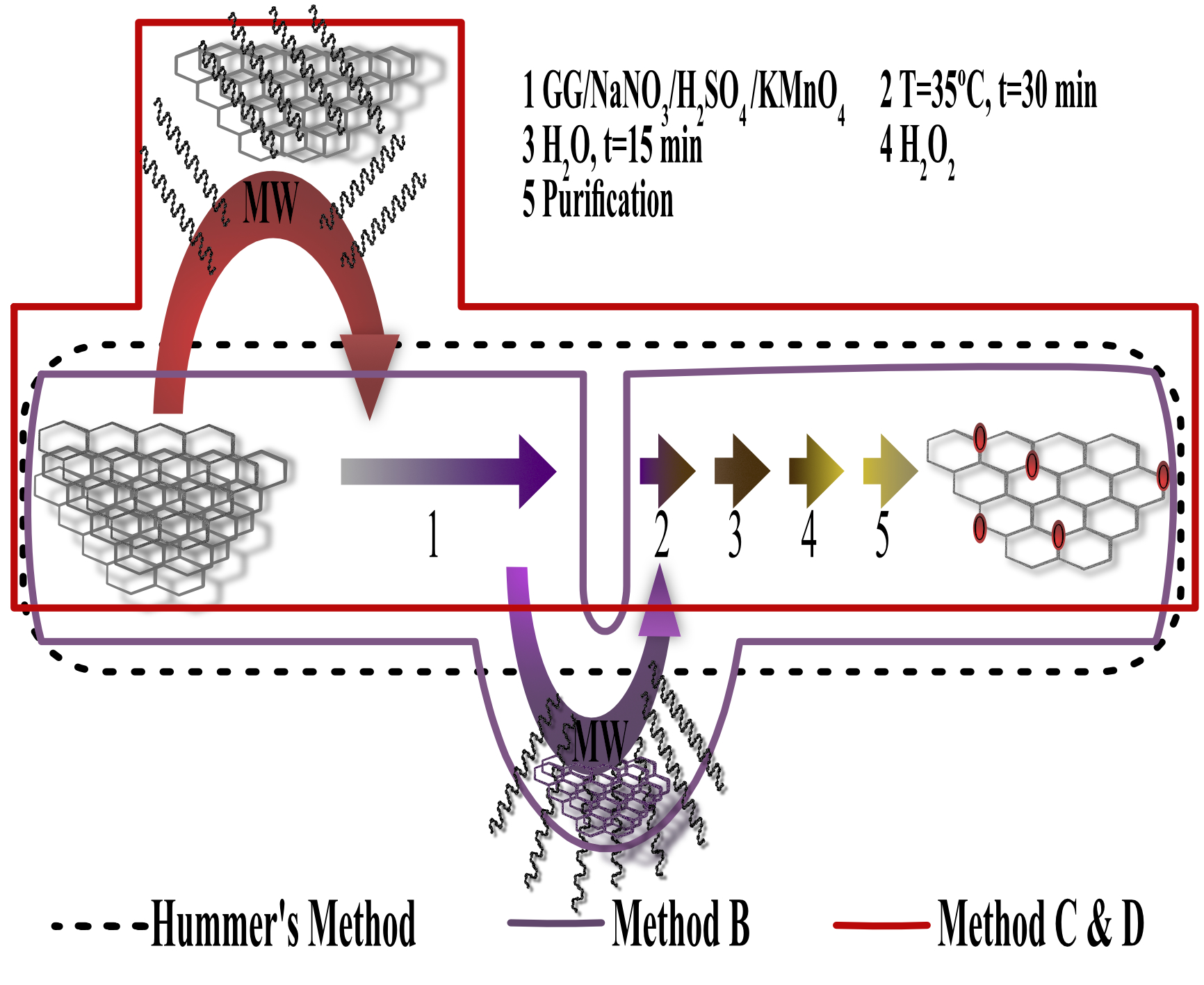
Nanomaterials | Free Full-Text | Synthesis of Graphite Oxide with Different Surface Oxygen Contents Assisted Microwave Radiation | HTML

MnO2 particles grown on the surface of N-doped hollow porous carbon nanospheres for aqueous rechargeable zinc ion batteries - ScienceDirect

Polypyrrole-Coated Sodium Manganate Hollow Microspheres as a Superior Cathode for Sodium Ion Batteries - ACS Appl. Mater. Interfaces - X-MOL

PDF) ChemInform Abstract: Beliefs and Facts in Permanganate Chemistry - An Overview on the Synthesis and the Reactivity of Simple and Complex Permanganates
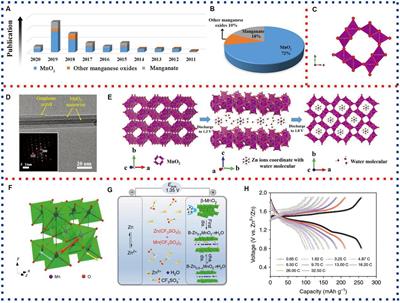
Frontiers | Application of Manganese-Based Materials in Aqueous Rechargeable Zinc-Ion Batteries | Energy Research
![A halogenophilic pathway in the reactions of transition metal carbonyl anions with [(η 6 -iodobenzene)Cr(CO) 3 ] - Dalton Transactions (RSC Publishing) DOI:10.1039/C4DT01363H A halogenophilic pathway in the reactions of transition metal carbonyl anions with [(η 6 -iodobenzene)Cr(CO) 3 ] - Dalton Transactions (RSC Publishing) DOI:10.1039/C4DT01363H](https://pubs.rsc.org/image/article/2014/DT/c4dt01363h/c4dt01363h-s5_hi-res.gif)
A halogenophilic pathway in the reactions of transition metal carbonyl anions with [(η 6 -iodobenzene)Cr(CO) 3 ] - Dalton Transactions (RSC Publishing) DOI:10.1039/C4DT01363H
Electron resonance studies of transition metal oxyions, I. Experimental results for the manganate, hypomanganate and ferrate ion
Electron resonance studies of transition metal oxyions, I. Experimental results for the manganate, hypomanganate and ferrate ion

PDF) Crystal structures of synthetic 7 Å and 10 Å manganates substituted by mono- and divalent cations
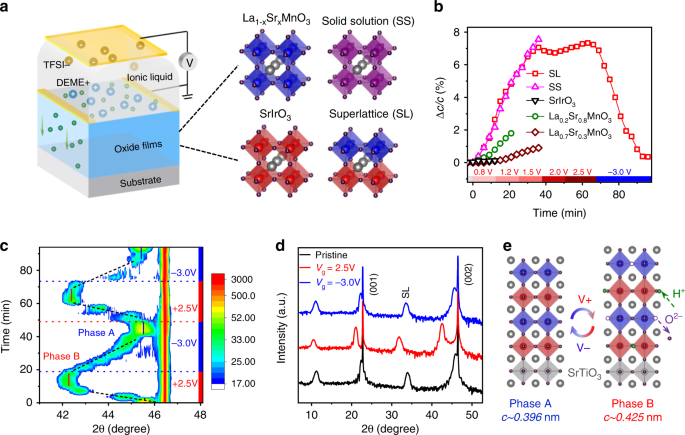
Emergent electric field control of phase transformation in oxide superlattices | Nature Communications

Schematics of the chemistry of the zinc-ion battery. Zn²⁺ ions migrate... | Download Scientific Diagram

PDF) Crystal structures of synthetic 7 Å and 10 Å manganates substituted by mono- and divalent cations
Electron resonance studies of transition metal oxyions, I. Experimental results for the manganate, hypomanganate and ferrate ion
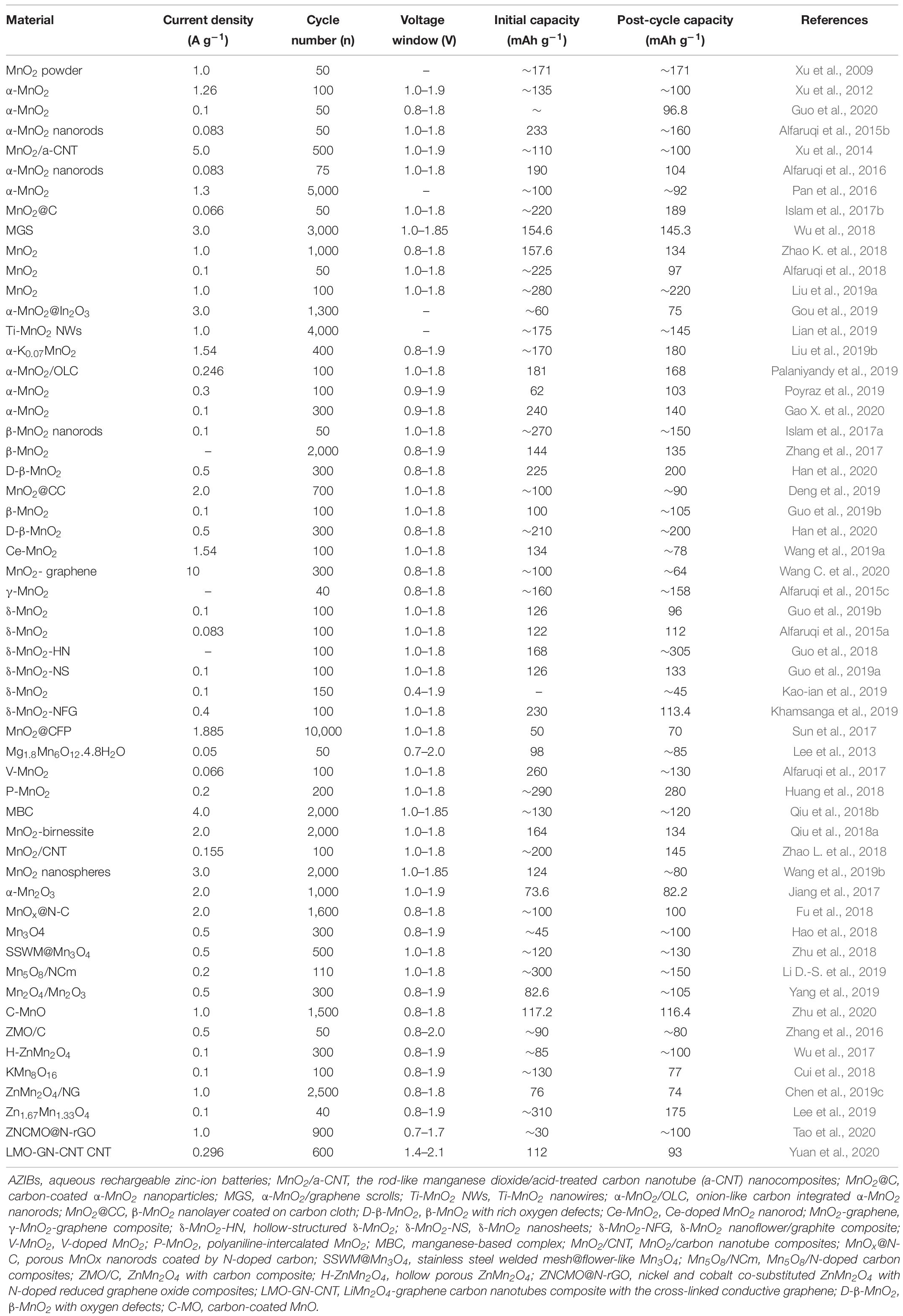
Frontiers | Application of Manganese-Based Materials in Aqueous Rechargeable Zinc-Ion Batteries | Energy Research

Fast Microwave-Assisted Hydrothermal Synthesis of Pure Layered δ-MnO₂ for Multivalent Ion Intercalation. - Abstract - Europe PMC

Kinetic analysis of the Li+ ion intercalation behavior of solution derived nano-crystalline lithium manganate thin films
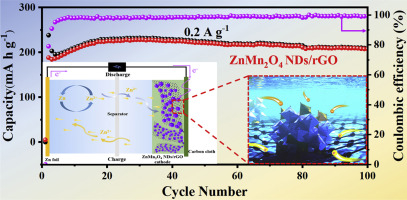
Strongly coupled zinc manganate nanodots and graphene composite as an advanced cathode material for aqueous zinc ion batteries - Ceram. Int. - X-MOL